Veterinary / PRODUCT
HOME>Product>Animal Health>Veterinary
Tiamulin Fumarate
Chemical Name | :Tiamulin Fumarate |
Category | :Antibiotics |
Specification | :CP/USP/EP |
HS Code | :29419090.00 |
- Tel: +86-532-83876123
- Fax: +86-532-83876157
- Email: dennis@qingmeibio.com
- Skype: dennis10221
Structure Formula | :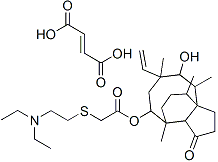 |
CAS Number | :55297-96-6 |
Molecular Formula | :C32H51NO8S |
Usage | :Mainly used for the prevention and treatment of chronic respiratory disease, swine mycoplasmal pneumonia (asthma), actinomycetes pleura pneumonia and treponema dysentery, etc.Low dose can promote growth, improve feed utilization rate. |
Items | Standard |
|---|---|
Appearance | White or almost white crystal powder;Have no smelly and tasteless |
Meting temperature | 143~152° |
Specific rotation | +24°~+28° |
Fumarate | 83.7~87.3mg |
Identification | HPLC:The retention time of the test solution corresponds to that of the standard corresponds to that of the standard solution. IR spectrum:IR spectrum of the sample corresponds to that of the reference standard |
pH | 3.1~4.1 |
Chromatographic purity | Not more than 1.0% of any identified impurity is found Not more than 0.5% of any unidentified impurity is foun And not more than 3.0% of total impuritied is foud |
Color and clarity of solution | ≤0.030(650nm) ≤0.150(400nm) |
Limit of residual solvents | 4-methyl-pentanone≤5000ppm methanol≤3000ppm benzene≤2ppm |
Loss on drying | ≤0.5% |
Residue on ignition | ≤0.1% |
Heavy Metals | ≤0.001% |
Assay | 97.0~102.0% |
1, Argentinian National Institute of Drugs
Instituto Nacional de Medicamentos (INAME)
2, Australian Therapeutic Goods Administration (TGA)
3, Belgian Federal Agency for Medicines and Health Products
Agence Fédérale des Médicaments et des Produits de Santé(AFMPS)
Federaal Agentschap voor Geneesmiddelen enGezondheidsproducten (FAGG)
4, Canadian Health Products and Food Branch Inspectorate(HPFBI)
5, Taiwan Food and Drug Administration (TFDA)
7, Czech State Institute for Drug Control
Státní Ústav pro Kontrolu Léčiv (SÚKL)
8, Czech Institute for State Control of Veterinary Biologicals and Medicines (ISCVBM)
9, Danish Health and Medicines Authority (DHMA)
10, Finnish Medicines Agency (FIMEA)
11, French National Agency for Medicines and Health Products Safety
Agence nationale de sécurité du médicament et des produits de santé (ANSM)
12, French Agency for Food, Environmental & Occupational Health Safety
Agence nationale de sécurité sanitaire de l'alimentation, de l'environnement et du travail (ANSES)
13, German Federal Ministry of Health *
Bundesministerium für Gesundheit (BMG)
14, Icelandic Medicines Agency (IMA)
20, Indonesian National Agency for Drug and Food Control (NADFC)
21, Health Products Regulatory Authority (HPRA)
22, Italian Medicines Agency
Agenzia Italiana del Farmaco (AIFA)
23, Japanese Pharmaceuticals and Medical Devices Agency(PMDA)
24, Korea (Republic of) Ministry of Food and Drug Safety (MFDS)
25, Malaysian National Pharmaceutical Control Bureau (NPCB)
26, Dutch Health Care Inspectorate*
Inspectie voor de Gezondheidszorg (IGZ)
27, New Zealand's Medicines and Medical Devices Safety Authority (Medsafe)
28, Norwegian Medicines Agency (NOMA)
29, Polish Main Pharmaceutical Inspectorate (MPI)
30, Romanian National Agency for Medicines and MedicalDevices (NAMMD)
31, South African Medicines Control Council (MCC)
32, Spanish Agency of Medicines and Medical Devices *
Agencia Española de Medicamentos y Productos Sanitarios(AEMPS)
33, Swedish Medical Products Agency (MPA)
34, United Kingdom's Medicines and Healthcare Products Regulatory Agency (MHRA)
35, United Kingdom's Veterinary Medicine Directorate (VMD)
36, U.S. Food and Drug Administration (US FDA)
37, European Directorate for the Quality of Medicines & HealthCare (EDQM)
38, European Medicines Agency(EMA)
39, World Health Organization(WHO)