Ciprofloxacin
Chemical Name | :Ciprofloxacin |
Category | :The third generation ofquinolone antibiotics |
Specification | :EP/USP |
HS Code | :30049090.90 |
- Tel: +86-532-83876123
- Fax: +86-532-83876157
- Email: dennis@qingmeibio.com
- Skype: dennis10221
Structure Formula | :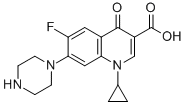 |
CAS Number | :85721-33-1 |
Molecular Formula | :C17H18FN3O3 |
Usage | :Ciprofloxacin is the synthesis of the third generation quinolone antibacterial drugs, with broad-spectrum antibacterial activity and good bactericidal effect. Its effect of antibacterial activity is 2~4 times stronger than that of norfloxacin and enoxacin for almost all bacteria.It has antibacterial effect on e. coli, pseudomonas aeruginosa, h. influenzae, neisseria gonorrhoeae, legionella bacteria, staphylococcus aureus, streptococcus. |
Items | Standard |
|---|---|
Appearance | White to off-white, ordorless,crystalline powder |
Identification | A: IR B: UV |
Specific optical rotation | +45~+51° |
Chromatographic purity Any individual impurity Total impurities | ≤1.0% ≤1.5% |
Ralated compound A | ≤0.5% |
Melting point | About 205℃ |
Loss on drying | ≤1.0% |
Solubility | Freely soluble in choroform; sparingly soluble in alcohol and in methanol;slightly soluble in ether; insoluble in water. |
Residual solvents Methanol Methylene chloride | ≤3000ppm ≤500ppm |
Assay | 97.0~103.0% |
Micron | 100%≤20μm |
Conclusion | Conform the stipulation |
1, Argentinian National Institute of Drugs
Instituto Nacional de Medicamentos (INAME)
2, Australian Therapeutic Goods Administration (TGA)
3, Belgian Federal Agency for Medicines and Health Products
Agence Fédérale des Médicaments et des Produits de Santé(AFMPS)
Federaal Agentschap voor Geneesmiddelen enGezondheidsproducten (FAGG)
4, Canadian Health Products and Food Branch Inspectorate(HPFBI)
5, Taiwan Food and Drug Administration (TFDA)
7, Czech State Institute for Drug Control
Státní Ústav pro Kontrolu Léčiv (SÚKL)
8, Czech Institute for State Control of Veterinary Biologicals and Medicines (ISCVBM)
9, Danish Health and Medicines Authority (DHMA)
10, Finnish Medicines Agency (FIMEA)
11, French National Agency for Medicines and Health Products Safety
Agence nationale de sécurité du médicament et des produits de santé (ANSM)
12, French Agency for Food, Environmental & Occupational Health Safety
Agence nationale de sécurité sanitaire de l'alimentation, de l'environnement et du travail (ANSES)
13, German Federal Ministry of Health *
Bundesministerium für Gesundheit (BMG)
14, Icelandic Medicines Agency (IMA)
20, Indonesian National Agency for Drug and Food Control (NADFC)
21, Health Products Regulatory Authority (HPRA)
22, Italian Medicines Agency
Agenzia Italiana del Farmaco (AIFA)
23, Japanese Pharmaceuticals and Medical Devices Agency(PMDA)
24, Korea (Republic of) Ministry of Food and Drug Safety (MFDS)
25, Malaysian National Pharmaceutical Control Bureau (NPCB)
26, Dutch Health Care Inspectorate*
Inspectie voor de Gezondheidszorg (IGZ)
27, New Zealand's Medicines and Medical Devices Safety Authority (Medsafe)
28, Norwegian Medicines Agency (NOMA)
29, Polish Main Pharmaceutical Inspectorate (MPI)
30, Romanian National Agency for Medicines and MedicalDevices (NAMMD)
31, South African Medicines Control Council (MCC)
32, Spanish Agency of Medicines and Medical Devices *
Agencia Española de Medicamentos y Productos Sanitarios(AEMPS)
33, Swedish Medical Products Agency (MPA)
34, United Kingdom's Medicines and Healthcare Products Regulatory Agency (MHRA)
35, United Kingdom's Veterinary Medicine Directorate (VMD)
36, U.S. Food and Drug Administration (US FDA)
37, European Directorate for the Quality of Medicines & HealthCare (EDQM)
38, European Medicines Agency(EMA)
39, World Health Organization(WHO)